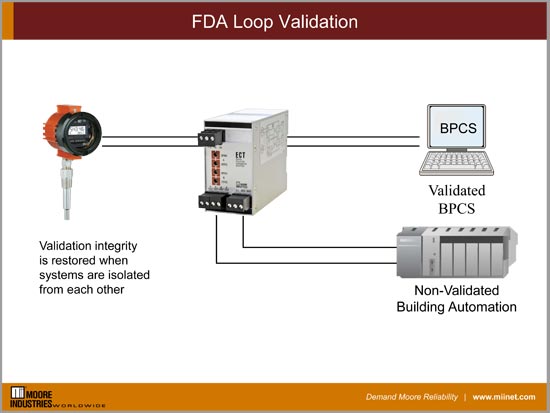
Food and beverage and pharmaceutical companies have several processes that require validation due to FDA and other international safety laws. When some measurements need to be shared between the primary process control system that is validated and an auxiliary control system, like building automation, it requires full isolation.
When this need arises using an ECT DIN Signal Splitter Isolator that provides the sufficient area isolation to meet validation needs. The UL approved ECT signal splitter now allows the non-validated system to be brought offline and calibrated separately without affecting the calibration or operation of the primary validated loop.
Key Attributes:
- Full four way isolation up to 500Vrms between power and all input/outputs
- RFI/EMI resistant and hardened aluminum case
- Zero and span potentiometers for onsite calibration